This post was originally published on this site Telix Pharmaceuticals has established a collaboration agreement with a team of researchers from the German Cancer Research Center (Deutsches Krebsforschungszentrum, or DKFZ) to develop an innovative image-guided surgery for men with prostate cancer. Investigators from the DKFZ and Heidelberg University Clinic have been working on the development…
Category: <span>Blog</span>
Expert Discusses Celsion’s Ovarian Cancer Immunotherapy GEN-1; Phase 1/2 Trial Still Enrolling
This post was originally published on this site The ovarian cancer immunotherapy candidate GEN-1, which is being tested in a Phase 1/2 clinical trial in newly diagnosed patients with advanced disease, was the focus of a recent call with a gynecologic oncology expert. The OVATION 2 trial (NCT03393884) is enrolling at 34 sites across the U.S. It…
GSK’s Belantamab Mafodotin Shows Promise for Relapsed or Refractory Multiple Myeloma
This post was originally published on this site GlaxoSmithKline’s investigational antibody-drug conjugate, belantamab mafodotin (GSK2857916), demonstrated a clinically meaningful overall response rate in hard-to-treat multiple myeloma patients in a Phase 2 trial, with the trial meeting its primary goal. Findings from the open-label DREAMM-2 study (NCT03525678) — which is assessing the treatment as a fourth-line…
$102.2M Pelotonia Gift to Help Establish Immuno-Oncology Institute at OSU
This post was originally published on this site A $102.2 million Pelotonia donation will help establish an institute at Ohio State University (OSU) that will focus exclusively on groundbreaking immuno-oncology therapies and cures for cancer. Disbursed over five years, the funds will support research to be conducted by the Pelotonia Institute for Immuno-Oncology (PIIO) at the…
USPSTF Updates Recommendations on Testing for BRCA-related Cancers in Women
This post was originally published on this site The United States Preventive Services Task Force (USPSTF) has revised their recommendations on testing for cancer-associated BRCA1/2 mutations in women, suggesting risk assessment only in those with a personal or family history of cancers associated with these mutations. These guidelines were published in the journal JAMA in a…
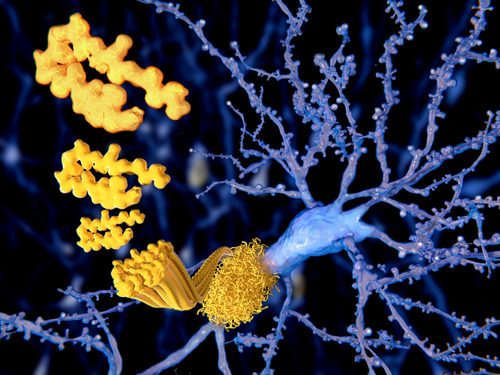
Understanding Protein Aggregation May Aid Quest for Better Alzheimer’s Treatments
This post was originally published on this site Better understanding the mathematics behind how protein clumps form — and how therapies interfere with this process — may improve treatment strategies for Alzheimer’s, and other diseases, new research suggests. The study, “Optimal control strategies for inhibition of protein aggregation,” was published in PNAS. Protein aggregation is the term…
Cirmtuzumab-Imbruvica Combo Trial Enrolling CLL, SLL Patients for Phase 2
This post was originally published on this site Following promising early efficacy results for a cirmtuzumab–Imbruvica treatment combo, a Phase 2 clinical trial has now been opened to people with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL). The opening of this randomized trial was based on the promising results seen in the prior Phase 1 stage, in…
EU Committee Recommends Orphan Drug Status for Ziopharm’s IL-12 Gene Therapy for Glioblastoma
This post was originally published on this site A European Medicines Agency (EMA) committee has recommended orphan drug designation for Ziopharm Oncology‘s interleukin (IL)-12 gene therapy — known as the Controlled IL-12 platform — for the treatment of glioblastoma, a very aggressive type of brain cancer. The European Commission will now decide whether or not to grant…
10 Commandments for Surviving Breast Cancer
This post was originally published on this site I hate giving advice, but I’ve learned lessons through my breast cancer journey that may be helpful to others in similar situations. After my diagnosis, a doctor told me I had three months to live. Against the odds, I survived, and along the way, I experienced profound…
Request for Xtandi to Treat Metastatic Hormone-sensitive Prostate Cancer Before FDA
This post was originally published on this site The U.S. Food and Drug Administration (FDA) has agreed to review Astellas Pharma and Pfizer‘s application for Xtandi (enzalutamide), requesting its approved use be expanded to include men whose metastatic prostate cancer still responds to hormone therapy. The supplemental new drug application, covering men with metastatic hormone-sensitive prostate…