A first patient has been dosed in a Phase 2 trial investigating the safety and long-term efficacy of bryostatin-1 in treating moderate and moderately severe Alzheimer’s disease, the therapy’s developer, Neurotrope, announced. The trial (NCT04538066), which is recruiting patients at several sites across the U.S., will build on data from previous studies assessing bryostatin-1. Specifically, it…
Conditions
Conditions
Tecartus Recommended for EU Approval as CAR T-cell Therapy for Mantle Cell Lymphoma
A committee of the European Medicines Agency (EMA) has recommended the conditional approval of Tecartus (brexucabtagene autoleucel) for the treatment of adults with relapsed or refractory mantle cell lymphoma (MCL). The recommendation by the Committee for Medicinal Products for Human Use (CHMP) for Kite Pharma‘s CAR T-cell therapy is specific for patients who received at least two…
Phase 1 Trial of SL-172154 in Treating Advanced Ovarian Cancer Opens
Shattuck Labs has opened a Phase 1 clinical trial to investigate its lead candidate, SL-172154, in treating people with advanced ovarian cancer, the company announced in a press release. The trial (NCT04406623) is recruiting up to 33 patients at three sites in the U.S.: the John Wayne Cancer Institute at Providence St. John’s Health Center in California,…
Provenge Improves Survival in mCRPC Patients Over Xtandi, Zytiga, Real-world Study Finds
Provenge (sipuleucel-T), by Dendreon Pharmaceuticals, is superior to Zytiga (abiraterone acetate) or Xtandi (enzalutamide) at prolonging the lives of men with metastatic castration-resistant prostate cancer (mCRPC) when added at any point in a treatment regimen, according to a real-life study in the U.S. The study, “A Retrospective Observational Analysis of Overall Survival with Sipuleucel-T in…
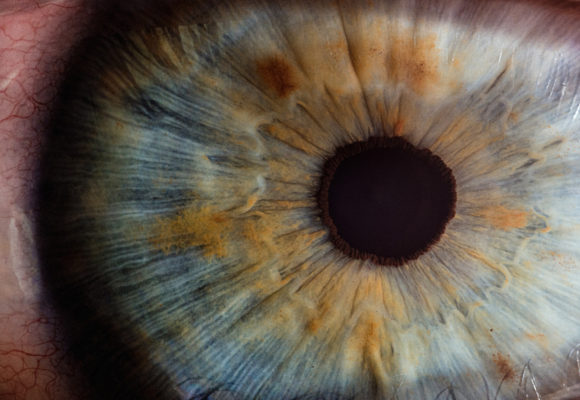
RetiSpec and Gentex Developing Technology to Detect Early Alzheimer’s
RetiSpec and Gentex are partnering to develop a technology to detect Alzheimer’s disease — potentially years before symptom onset — using a non-invasive and cost-effective eye scan technique. The partnership between the two companies will expedite the development of the technology, with the goal of bringing it to market to advance patient care. “The Gentex-RetiSpec partnership…
Committee Recommends EU Approval of Opdivo to Treat Advanced Esophageal Cancer
A committee of the European Medicines Agency has recommended the approval of Opdivo (nivolumab) for the treatment of people with advanced, inoperable, recurrent, or metastatic esophageal squamous cell carcinoma (ESCC) who previously received a chemo regimen containing fluoropyrimidine and a platinum agent. The European Commission will review this opinion from the Committee for Medicinal Products for Human…
Sarclisa Triple Combo Conditionally Approved for NHS Use in England, Wales
After first rejecting a Sarclisa (isatuximab) triple combination therapy due to cost-effectiveness concerns, the U.K.’s National Institute for Health and Care Excellence (NICE) has given conditional approval to the treatment for relapsed or refractory multiple myeloma. The triple therapy includes Sarclisa in combination with Imnovid (pomalidomide, also sold as Pomalyst) and dexamethasone, a corticosteroid. It is particularly recommended for…
COVID-19 Surgery Delays May Mean 2,700 More Breast Cancer Deaths Within 10 Years
Delays in breast cancer surgery due to the COVID-19 pandemic could cost 2,797 additional deaths in the U.S. over the next decade, according to a Kantar Health analysis. Such treatment delays also place patients at a higher risk of metastasis, or cancer spread, potentially increasing healthcare costs in the U.S. by an estimated $376 million…
FDA Places PC14586, for Cancers with Mutant Protein, on Fast Track
The U.S. Food and Drug Administration (FDA) has granted fast track designation to PC14586 as a potential treatment of advanced cancers that harbor a specific mutation affecting the protein p53, which includes some ovarian cancers. Fast track is given to support therapies with the potential to fill a serious unmet medical need. They are eligible…
ADT Not Needed With Radiation in Favorable Intermediate-Risk PC
Adding four months of androgen deprivation therapy (ADT) to radiation treatment extends survival and delays metastasis in men with unfavorable intermediate-risk prostate cancer, according to a secondary analysis of a Phase 3 clinical trial. However, that new trial analysis showed there were no such benefits to ADT therapy for men with favorable intermediate-risk prostate cancer undergoing…
Cancer Vaccine UV1 Plus Keytruda Shows Safety, Early Efficacy in Advanced Melanoma Trial
Adding UV1, Ultimovacs’ experimental cancer vaccine, to Keytruda (pembrolizumab) is safe and shows initial signs of effectiveness in adults with previously untreated metastatic melanoma, according to top-line data from a Phase 1 clinical trial. “The safety profile observed to date in this first cohort [group of patients] of the [UV1-Keytruda] combination study is consistent with the promising…
Oncopeptides Will Seek Conditional Approval of Melflufen in Europe
Oncopeptides soon will seek conditional approval of melflufen (melphalan flufenamide) in the EU for the treatment of relapsed or refractory multiple myeloma. The pharmaceutical company now is planning to submit its application — some two years earlier than expected — to the European Medicines Agency (EMA) to request melflufen’s conditional approval. The request was originally intended to…