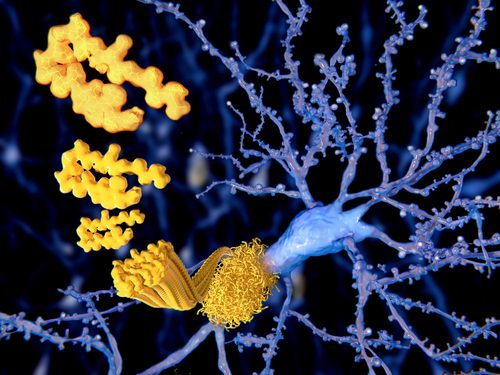
Eisai and Biogen’s experimental therapy lecanemab (BAN2401) safely leads to rapid and sustained reductions in the brain levels of beta-amyloid — the protein that forms toxic clumps in Alzheimer’s — in people in early disease stages, according to one-year, preliminary data from a Phase 2 trial’s ongoing extension phase. Notably, greater beta-amyloid drops were observed in…
Conditions
Conditions
#AANAM – EMBARK Trial to Again Test Safety, Efficacy of Aducanumab
A new Phase 3 clinical trial, called EMBARK, will evaluate the long-term safety and efficacy of aducanumab (BIIB037), an investigational therapy for Alzheimer’s disease in patients who took part in its previous clinical studies. The trial’s design was described by Carmen Castrillo-Viguera, a medical director at Biogen, in the poster “EMBARK: A Phase 3b, Open-Label, Single-Arm, Safety…
Movement Gets Youth Involved in Alzheimer’s Advocacy, Education
Alzheimer’s disease is not often seen among those younger than 65, but the Youth Movement Against Alzheimer’s is getting young people involved with the neurological disorder through volunteer work, education, and advocacy. While they aren’t typically affected themselves, young people often are left caring for a family member with the disease. They’re also indirectly burdened…
#AANAM – Nuplazid Not Linked to Side Effects Common to Antipsychotics
Acadia Pharmaceuticals’ Nuplazid (pimavanserin) — which is being evaluated as a potential treatment for hallucinations and delusions associated with dementia-related psychosis — is not associated with common motor side effects of conventional antipsychotics, according to an analysis of data from three clinical trials. These findings add to previous Phase 3 trial results showing that the…
FDA Places Gene Therapy LX1001 on Fast Track
LX1001, a gene therapy for Alzheimer’s disease being developed by Lexeo Therapeutics, has been granted fast track designation by the U.S. Food and Drug Administration (FDA). The therapy is designed to deliver a version of the APOE gene, called APOE2, to cells in the central nervous system (the brain and spinal cord) using an engineered…
Longeveron’s Stem Cell Therapy Shows Potential in Phase 1 Trial
Longeveron’s Lomecel-B, an investigational bone marrow-derived medicinal signaling cell (MSC), showed an ability to slow cognitive decline and the loss of daily life abilities in patients with mild Alzheimer’s disease relative to those given a placebo in a Phase 1 clinical trial, the company announced. Treatment with Lomecel-B was deemed safe, with the therapy meeting the trial’s primary safety…
Insufficient Evidence of Aducanumab Efficacy, ICER Draft Report Finds
A draft report from the Institute for Clinical and Economic Review, known as ICER, found that there is insufficient evidence to determine whether the investigational therapy aducanumab will provide health benefits to people with Alzheimer’s disease. ICER is an independent non-profit research institute that works to assess the effectiveness and value of medicines and other…
$10.7M NIH Grant to Fund Genetic Research in Alzheimer’s
A $10.7-million grant to investigate the genetic underpinnings of Alzheimer’s disease has been awarded to the University of Pittsburgh Graduate School of Public Health and Washington University School of Medicine in St. Louis. Despite years of research, scientists have yet to come up with a cure for Alzheimer’s disease — or a way to prevent…
Annovis Seeks Orphan Drug Status for ANVS401 for Down Syndrome Patients
Annovis Bio is seeking orphan drug status for its investigational therapy ANVS401, known as Posiphen, for treating Alzheimer’s disease in people with Down syndrome (DS-AD). The clinical-stage drug platform company has filed an application with the U.S. Food and Drug Administration (FDA) for such designation, granted to therapies for rare diseases. “The FDA orphan drug application…
Brown University to Focus on Early Diagnosis, Treatment
Brown University is establishing the Center for Alzheimer’s Disease Research to build a world-class research program that is focused on the early detection and treatment of Alzheimer’s disease and related disorders. The center is made possible by separate gifts of $25 million and $5 million from donors who wish to be anonymous. It will bring together…
Association Endorses ENACT Act to Improve Clinical Trial Diversity
The Alzheimer’s Association and its advocacy affiliate, the Alzheimer’s Impact Movement (AIM), are backing recently introduced bipartisan legislation to support equity in Alzheimer’s disease clinical trials. The Equity in Neuroscience and Alzheimer’s Clinical Trials (ENACT) Act of 2021 (H.R. 3085/S. 1548) would amend the Public Health Service Act to improve the diversity of participants in…
ProMIS, Selexis to Develop Antibody Targeting Toxic Amyloid Beta
ProMIS Neurosciences will advance the development of PMN310, its manmade antibody designed to target the toxic forms of amyloid beta that underlie Alzheimer’s disease. The first step in the manufacturing of PMN310 will be carried out by Selexis with its proprietary CHO-K1 cell line, part of the company’s SUREtechnology platform. The platform uses Selexis’ optimized, high-performance mammalian (Chinese…